MEET THE INVESTIGATORS
How reporting on the medical device industry has changed, five years after Implant Files
ICIJ member Jet Schouten kicked off the 2018 project with her reporting on lax regulation in the Netherlands. Since then, reporting on medical devices has only become more difficult.

The International Consortium of Investigative Journalists collaborates with hundreds of members across the world. Each of these journalists is among the best in his or her country and many have won national and global awards. Our monthly series, Meet the Investigators, highlights the work of these tireless journalists.
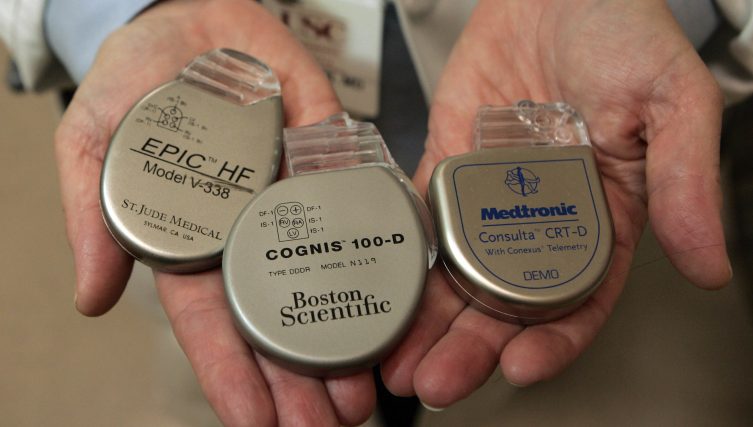
Five years ago, ICIJ published the Implant Files, a 260-journalists strong collaboration that exposed the flaws of the $400 billion global medical device industry.
The global investigation documented how regulators around the world approved devices — such as dental implants, surgical staplers, respirators and more — with little or no safety testing. And when those devices were found to be faulty or recalled, patients were often left out of the loop.
Implant Files, which spanned 36 countries and required the analysis of more than eight million device-related records, first began to take shape when ICIJ member Jet Schouten exposed the hidden epidemic of medical-device malfunction in the Netherlands.
Schouten won awards for her undercover reporting that revealed how easily untested medical devices were approved by Dutch regulators. But now, five years later, Schouten says reporting on the industry has only become more difficult.
TRANSCRIPT:
Carmen Molina Acosta: Hello and welcome back to Meet the Investigators from the International Consortium of Investigative Journalists. I’m your host, Carmen Molina Acosta. If you’re joining us for the first time today, Meet the Investigators is a podcast, where we hear from ICIJ journalists from across the globe.
Today, we’re taking a look back at the Implant Files — a groundbreaking investigation into the medical devices industry that ICIJ published five years ago. To commemorate that anniversary, I sat down with the journalist who started it all.
Jet Schouten: My name is Jet Schouten and I’m a Dutch journalist. I work for the investigative desk of the Dutch newspaper NRC. I started Implant Files together with ICIJ.
Carmen: Back then, Jet was working for a TV program in the Netherlands, on a show called Radar.
Jet: It’s very popular here in the Netherlands, and the way the TV program works is that they respond to questions and letters that consumers have about really anything that has an impact on their lives. So my beat is health, so I was reading through the letters of people who were reporting health problems and health issues. And then I think it was around 2012 when I first received a letter from a woman who had a [transvaginal] mesh implanted in her and she had this terrible pains and ailments and the doctors could not help her at all.
Carmen: The mesh implanted in the woman, Jet says, was meant to treat a prolapse.
Jet: Which is actually really common, but you don’t really hear a lot about it because it’s a condition of people are usually ashamed of because it means that usually after childbirth, for example, your uterus or your intestines are coming through your vagina.
Carmen: Big manufacturers, like Johnson & Johnson, came up with what they thought was a solution.
Jet: They invented a plastic implant which has these arms. And with the arms, they secure it in the pelvic area. It’s a severe operation. And the purpose of this implant is that all the organs stay in place and fibrotic tissue in the pelvic area will create sort of a hammock through the implants.
So this was the idea, but the outcome was terrible because the plastic could protrude through the organs, creating this horrible effect on these women. And the worst part is that it’s impossible to take the implant out.
Carmen: Online, Jet found women all over the world with the same story. But when she reached out to the Dutch government, asking if they knew about the problem, they said they hadn’t gotten any complaints.
Jet: Radar is a very creative TV program. So, we went undercover and we made up our own vaginal mesh made out of a mandarin bag. So we went to the grocery store, we bought a bag of mandarins, we discarded the mandarins and we cut it up, we made some pictures. And then we went undercover with a hidden camera and we said to these regulators, like, “Okay, we want to sell this device to these women.” And mind you, it’s a big market, because one in four women will experience a prolapse in her life.
And then to our big surprise, the regulator said, “Well, we don’t see any problems if you want to sell this device.”
Carmen: That mandarin net kicked off the start of years-long investigations into medical devices. Jet’s colleague, ICIJ member Joop Bouma, reached out to ICIJ with the idea, and the Implant Files was born.

Regulating medical devices, it turns out, is a patchwork industry. Implant Files revealed there are all sorts of ways dangerous products can fall through the cracks.
Governments don’t always require stringent testing before devices go on the market, the way they might with a drug. Like the case of the mandarin net, faulty implants can get through if something vaguely similar is already being sold. And when there are problems, governments also don’t always communicate — meaning devices banned in some countries might still be for sale in others.
Jet: The regulatory oversight is very, very poor. And when I talk to the colleagues in the US, they really complain about the FDA, because they say, “Yeah, we have a database with medical device reports. But there is under reporting and it’s not complete and the FDA is not doing enough.” And then I say “Look, look at Europe!” In Europe, we don’t have a regulatory body at all. There is not a regulatory overarching body that sees what’s going on with medical devices in Europe.
That is very difficult to understand, but that’s how it is. So every European country has their own government, and the government looks at the devices in that particular country. So here the information transparency is very, very poor. Because if I want to find out what’s going on with device in my country, or in any other country in Europe for that matter, I have to submit a freedom of information request. That’s so different than in the States, because there you can go to the FDA. And I know that the FDA database is poor, but at least there is a database and at least there is awareness. So that, that’s a huge difference.
Carmen: Using that FDA database, Jet worked with ICIJ’s data journalists to track down the history of specific devices, and show how those regulatory gaps leave real patients vulnerable.
Jet: I started working together with a few people on looking for devices that had certain criteria. For example, they were used on a global scale; it was already known there were problems with these devices; they are high-risk devices, so something that’s being implanted into a body, and; it’s something that we can follow in time. Meaning, it’s on the markets already for a while so we can see how it evolves.
So, luckily ICIJ has this wonderful data team and we started working together scanning for patterns, looking for which devices cost the most harm. And also: what was done? Like, who knew what, and when?
And, yeah, if you start to look for these patterns, you usually see the same stuff. So you see that people are being injured. You’re seeing the type of injury, you’re seeing people die. And then you can look for: Okay, was action being done after that? So, was there a recall? Was there some notice sent to patients, to doctors? And then usually you see a lag time.
And these patterns slowly started to emerge. And once you see the patterns, you can say, “Okay, this is not a coincidence. It’s not just a device here or a device there, but this is a pattern and it means it’s a systemic problem.” And that’s what we did.

Carmen: A lot of those devices that Implant Files tracked were marketed to women — things like the mesh, contraceptive coils and breast implants.
Jet: These stories never leave you. I talked to a woman who started having breast implants, because her doctor recommended it after she had a mastectomy because she had a gene that made her susceptible for cancer.
But what she didn’t know is that, after a mastectomy, the chances of a rupture of breast implants are actually higher. So, she got a rupture of her implants and she didn’t know that the silicones were leaking into her body and then she got breast cancer, because of the silicones.
You know, you carry these stories with you and you want to help and you want to do something, you want to write about it. That’s the only thing that we can do as journalists. So that the regulator will say, “Hey, you know what? We need to change this.”
Carmen: But while Implant Files brought attention to these systemic issues, there’s still lots of regulatory work to be done. If anything, Jet says, investigating devices in Europe is harder today than it was 5 years ago. New regulations have made it much more difficult to get certain documents from government agencies, particularly when it comes to communications between regulators and states about problems with a device.
Jet: For example, I submitted the Freedom of Information requests to the Dutch government, and then the government didn’t give me the documents that I asked for.
So, I went to court and then the judge said, “Okay, you have to provide these documents,” they told the Dutch government. But it didn’t happen and instead they had to pay a fine.
So this is a relatively new thing, that fines are being paid to journalists. So instead of getting documents, we’re getting money, which is not what you want as a journalist. You don’t want the money, which is taxpayers’ money — of course, you want the document.
Carmen: Jet didn’t set out to be a journalist originally — a test she took in high school persuaded her to study theology.
Jet: I had a lot of different interests and I didn’t know what to study. And I was like: theology, really? Isn’t that something with Bibles and for religious people? So I was, I was really flabbergasted by that outcome. And then I started talking to people and I really became more and more drawn to this study. You have the chance to also study languages and philosophy and history, so it’s a very broad study and it’s exactly what I was looking for, I was interested in many things and that study had it all.
And then I thought, “OK, so what next?” and I stumbled upon an old friend and she told me that she worked for public radio. And she studied theology as well. So we started talking and I said I should come to see your work. And so we met up and I talked to her boss and one thing led to the other and then I started working in radio as well.
Carmen: There’s a certain tenacity you need to be a reporter, she says… which is how Jet ended up making the jump to working in television.
Jet: This was a wild jump because I responded to the ad in the newspaper. And then the ad specifically said, you only could apply for this job if you had TV experience. So I phoned them up and I told them “Look, I don’t have TV experience. But I like your show. It’s a consumer angle. I see this angle. It has a lot of overlap with what I’m doing for radio. So yeah, give me a shot. I think I can do it.”
And then I remember the chief editor told me on the phone that I would have less than 1% chance if I would write a letter to them. So … I wrote a letter to them.
Carmen: Not only did Jet apply — she put together a full 40-minute segment, with all the topics and interviews she would want to see on the program.
Jet: I thought that it was just a kind of a ridiculous answer, “Like, less than 1% chance.” And there is something that I have, and I think other journalists have as well, that if you say to a journalist, “I give you a very, very low chance that you will succeed” — then I really want to succeed. So I will try anything to make it happen, you know?
Carmen: Trying new things is not an unusual undertaking for Jet … after Implant Files wrapped up, she traded the newsroom for a classroom and accepted a year-long Knight Wallace fellowship at the University of Michigan.
Jet: I think it was the best thing I could have done after Implant Files, because it gave me a chance to really catch my breath and to think about what to do next. The program is designed in a way where they say to the fellows. “Look, you’re all accomplished journalists. You were thinking all about what to do next and in order to help you with that, we want you to choose one course at the University of Michigan (and we had a free pass to take any class there). We want you to take a class that is completely, but then also utterly out of your comfort zone.” And they call it your crazy course.
Carmen: So, she took courses on African dance, and learned about the largest musical instruments in the world — the carillon, huge towers filled with multiple bells that can be heard for miles around.
Jet: It’s a very peculiar instrument because it’s, I think, the only public instrument in the world, which means if you play it, people hear it. There is a connection with the community. You have a connection with the audience, which are the people on the street.
Carmen: The pandemic cut the fellowship short, but Jet went back to the Netherlands and continued to learn about carillons. After all, bells are an instrument that you could still play — and listen to — publicly.
Jet: I remember reading something about in the Middle Ages, there was a superstition about the healing aspect of bells. So people thought that ringing the bells had a sort of a healing aspect to it, for example, when there was the plague, the bells would ring. And it was a belief that this could help cast the plague away in the city. Now [coronavirus] just started, so I talked with Pamela, my teacher, about it and we started composing from this idea a piece around the Covid virus that was just known to the world.
Carmen: They called it Healing Bells.
Jet: The day before I went home, we finished writing this piece. And then the piece was performed in several countries a few weeks after that, on several carillons around the globe. People could open their windows in their home and hear the music, and we were hoping to spread a message of hope through the sounds of the bells and that’s what we did.
It’s very great to do something that you haven’t done before. There is something very, very empowering and discovering in your forties that there is something hidden inside of you that you didn’t know you had a passion for, and you didn’t know you loved doing so much. And that is just wildly inspiring.
You know, as journalists, we can write stories on the same topics again and again, particularly when you look at medical devices because so little changes in the problems and stakes are big and high. But then it’s equally important to recharge and to replenish and to open new doors inside of you and start to think in a different way.
Carmen: Recently, Jet worked with a team of reporters at ProPublica and the [Pittsburgh Post-Gazette] on an investigation into Philips Respironics, which makes ventilators and CPAP machines. They found that for over a decade, Phillips hid thousands of reports that an industrial foam in the devices was leaking toxic and carcinogenic particles.
The project is another lesson in collaboration — and the strength journalism has to hold power to account.
Jet: Don’t take anything for granted. When I hear something or read something, I think of: who benefits from this? Who wins because of this, and who loses because of this? And it can make immediately something clear.
If you have the passion, your passion will lead you there, and you just want to know. You just want to know how some things are possible and how some things are construed, and how it was thought of, and what happened and who knew something and why didn’t they say anything and where are the documents? And you just get this inspiration to start digging in. And once you feel the inspiration to do it, then you do it. And I think the best advice I can give to journalists is to just follow their passion. And the rest will come.
Carmen: That was ICIJ member Jet Schouten. You can read stories from Implant Files, and any updates at icij.org. I’m Carmen Molina Acosta, and that’s it for this episode of Meet the Investigators. Thank you for listening! We’ll see you next time.
Correction, Dec. 22: This article misstated the investigation into Philips Respironics as a collaboration between ProPublica and the Philadelphia Inquirer. The project was a collaboration with the Pittsburgh Post-Gazette.


