After Bhinnata Piya’s mother was diagnosed with esophageal cancer in 2020, Piya was determined to be a supportive caregiver. Her father had died of the same type of cancer only three years earlier, and the loss devastated the tight-knit Nepalese family.
“When our dad died, I was just crying so much. We didn’t do enough,” Piya told the International Consortium of Investigative Journalists. “But when my mom died, I really felt like we did all that we could.”
Piya had moved back to Nepal from Cleveland for six months while her husband, Dain Finke, an American physician, remained in the United States. There she cared for her 63-year-old mother, Sita Gurung, during the day and worked remotely as a project manager for a health care technology company at night, until she could no longer manage it all.
Piya quit her job. She paid thousands of dollars toward Gurung’s more than $40,000 treatment. She accompanied her to India for medical scans. She even watched dutifully as Gurung, increasingly frail and eventually unable to eat, spent hours in the kitchen teaching her how to cook her favorite recipes.
“She once told me that if it weren’t for me, she would have died a long time back,” Piya, now 36, said.

At the end of 2022, as Gurung’s health worsened, her doctor prescribed an expensive immunotherapy treatment called Keytruda. Following the doctor’s recommendation, they bought the medication from a hospital worker in India who ran a medical tourism business.
Gurung was on the medication for two months, but it made no difference.
On Feb. 9, 2023, after enduring 2½ years of grueling chemotherapy, radiation and immunotherapy, she died at home with her elder daughter and Piya by her side.
“I took peace in knowing that we did everything,” Piya said.
But that feeling was short-lived.
One year after Gurung’s death, Piya’s sister learned shocking news while visiting her in Ohio and told her, “You need to sit down for this.” An Indian newspaper had reported that the man who sold them their mother’s cancer medication had been arrested. Police in New Delhi charged him and several others with selling adulterated drugs after allegedly finding they had filled vials labeled as Keytruda or other expensive oncology treatments with antifungal medicine, according to police records.
Piya never tested her mother’s medication, never suspected it could be fake. But the news planted a terrible fear in her mind: that she had possibly given her mother fake drugs.
“I just felt so numb,” Piya said.




For people with certain cancers, Keytruda is a lifeline. It can extend some patients’ lives by years or even cure them entirely.
Produced by the pharmaceutical giant Merck & Co., the blockbuster drug is used to treat more than a dozen types of cancer, including types of lung and breast cancers. But the cost can be ruinous.
A 200 milligram dose of Keytruda ranges from $1,700 in Indonesia to $12,000 in the U.S., according to an ICIJ analysis of recent list prices. Typically administered via intravenous infusion every three weeks, treatment can last up to two years — amounting to a staggering $416,000 in the U.S. before discounts.
In 2025, Keytruda accounted for nearly half of Merck’s $65 billion revenue, making it the company’s biggest moneymaker. And Merck has worked tirelessly to keep it that way.
The Cancer Calculus, a new global investigation by ICIJ and 47 media partners, found that Merck has exploited the global patent system to ward off competitors. It has also promoted a higher dosage of Keytruda than is often necessary, driving up costs for patients and hospitals, in its aggressive campaign to boost revenue.
The resulting high prices have contributed to vast disparities in access depending on where patients live and how much they, their governments or their health insurers are willing or able to pay. This dynamic has created a dangerous opening: new opportunities for counterfeiters to cash in on demand for costly oncology medicines as cancer rates soar worldwide.

ICIJ and its partners filed dozens of public records requests, interviewed investigators and experts, approached illegal sellers, and found that not only patients — but hospitals — have become unwitting customers of counterfeiters.
One patient in Mexico told broadcaster Univision that he was administered fake Keytruda in a public hospital. His account was among several alarming incidents that ICIJ identified in the country, where at least one person died after being “infused” with counterfeit Keytruda, according to Merck.
Anthony Zook, Merck’s associate vice president for global security, told ICIJ in an email that “criminal groups are now more commonly targeting life-saving medicines.”
“This shift is financially driven,” he said. Even so, in a statement to ICIJ, Merck defended its pricing.
“We have a long history of responsibly pricing our medicines to reflect their value to patients, payers and society,” said Johanna Herrmann, senior vice president and chief communications officer at Merck.
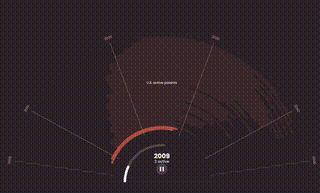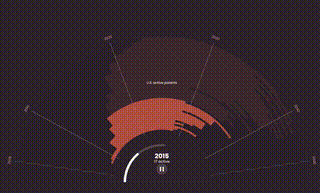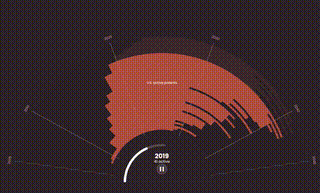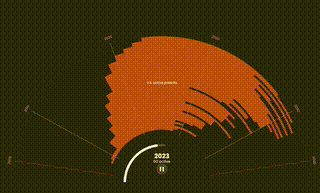
The World Health Organization (WHO) predicts that by 2050, cancer rates will surge to over 35 million cases, a 77% increase from 2022, disproportionately affecting patients in lower-income countries. These are the same people who already have the least access to basic health care, let alone expensive oncology treatments like Keytruda.
For counterfeiters, it’s all upside. “It probably cost them 10 bucks to make it when they’re selling it for $1,500, $2,000, $4,000,” said Kris Buckner, the founder of Investigative Consultants, a California-based private investigations firm that tracks down counterfeit products, including pharmaceuticals. Plus, if a cancer patient dies, it’s unlikely anyone will ever know whether they died from cancer or from taking a fake drug. “It’s the perfect crime.”
As long as Keytruda is priced like a luxury good, it will inevitably spawn cheap imitations and drive counterfeiters to exploit vulnerable people.
A ‘rational’ alternative
Keytruda, known generically as pembrolizumab, weaponizes a patient’s immune system against cancer cells. Unlike chemotherapy, which targets rapidly dividing cells, Keytruda blocks the PD-1 receptor on immune cells from binding with a protein that can prevent the body from identifying and attacking cancer.
“It’s been a game changer in the way we manage cancer, and also the outcomes of patients have improved substantially for many, many cancers,” said Binay Shah, a hematologist and oncologist who works in the U.S. and Nepal and co-founded the Binaytara Foundation, an organization focused on improving health care access.
But Keytruda’s success has come amid what the WHO called an “alarming” rise in the number of reported incidents of substandard and falsified medical products around the world in recent years. (Falsified products deliberately misrepresent their identity, composition or source, whereas substandard products fail to meet quality standards.) Since 2019, the WHO has recorded 36 product reports of falsified Keytruda, each corresponding to “several thousand doses or just a few,” according to a spokesperson. But the numbers likely capture only a fraction of the problem, in part because it’s easy to overlook negative reactions to falsified drugs, particularly in cancer patients.

Falsified drugs spread in several ways. Counterfeiters may conspire with unscrupulous hospital or pharmacy employees to steal authentic packaging, like Keytruda vials, and refill them with other substances, according to Europol, the European Union’s law enforcement agency. Counterfeiters can also fake packaging. To an untrained eye, these products look authentic but have made-up ingredients or batch numbers — the combination of letters and numbers that manufacturers use to identify and trace shipments. Such products are often sold on social media platforms, messaging apps like WhatsApp or online marketplaces where patients seek cheaper alternatives. The WHO spokesperson said that “significant volumes” of these sophisticated knockoffs “may be in circulation.”
“Consumers are actively looking for medications outside of the controlled supply chain,” said Tim Mackey, a University of California, San Diego, professor and public health researcher who studied and wrote about how counterfeit versions of the cancer medication Avastin penetrated the U.S. supply chain in 2012.
The search for affordable medicine can inadvertently lead people to one of the tens of thousands of online pharmacies operating illegally, or even to a storefront run by rogue sellers or organized crime groups. In both cases, the dangers may not be immediately clear.
One such storefront is in El Santuario, a neighborhood in the heart of Guadalajara, Mexico. The main road — ironically named Hospital — is overrun with illegal pharmacies. Instead of wearing white coats, the boys and men posing as pharmacists wear flat-brimmed hats.

“What are you looking for, man?” one of the pharmacists, a burly man with a teardrop tattoo, asked a reporter for ICIJ’s partner El País who visited the neighborhood in December.
“Keytruda,” the reporter responded.
“Keytruda?” he called out to a colleague inside the store.
“Give me a second,” the colleague said. He searched on his phone and found one 100 mg vial in stock. It cost 30,000 pesos (nearly $1,700) — roughly one-third the price advertised online by pharmacies in Mexico.
Buying medication in an environment like this or online may seem risky, but for cancer patients who cannot afford or do not have access to a potentially lifesaving drug, sourcing it wherever they can is a “pretty rational thing,” Mackey said. Sometimes, it’s the only option.
Seeking Keytruda
Once, when she was a college student in central Nepal, Sita Gurung passed a campus bulletin board and noticed someone had pinned a poem to it. She later met the author, Mahendra Piya, a fellow student and the man who would become her husband.
“The story is that she fell in love with the poet before my dad,” Bhinnata Piya said.
The couple came from different castes and social classes, which made them an unlikely match. “They eloped to Kathmandu because their families did not approve of their relationship or marriage,” Piya said. They carried that unorthodox streak into their marriage as he became a stay-at-home dad and she took a corporate job at Nepal Airlines, working her way up from accounts officer to finance executive.
“It was very rare at that time for a woman to work, let alone be successful,” Piya said. “She was fiercely independent for a Nepali woman.”

In 2016, Mahendra Piya was diagnosed with esophageal cancer, and his health deteriorated rapidly. About a year into his illness, Bhinnata Piya’s sister called to tell her she needed to come home. Soon after Piya and her husband arrived in Nepal, they awoke in the middle of the night to find her father losing consciousness. They raced toward the hospital with Piya cradling her father in the back of the car, but he died in her arms before they arrived.
“For a year it was just like a repeat of that scene over and over again in my head,” Piya said.
Gurung mourned the death of her husband, but her life slowly began to open up in new directions. In her 60s, she decided to pursue a postgraduate degree in counseling psychology.
Around that time, Gurung, who had always been health-conscious, became fearful that she too would get esophageal cancer. Piya thought her mother was being paranoid, but one day “she just called me, maybe after three years, and was like, ‘I’m at the hospital and I have what your dad [had]. And they’re saying I’m going to die basically because of the cancer,’ ” Piya recalled. Gurung had already started chemotherapy. “It felt unreal.”

Piya helped transfer her mother from the hospital where her father had been treated to a state-of-the-art facility about 10 miles south of Kathmandu, the nation’s capital. At first, she was impressed by Gurung’s new oncologist, Pankaj Barman, who seemed professional and respectful and actually listened — something her father’s doctors failed to do, Piya said. Barman put Gurung on a treatment plan that included chemotherapy and radiation, and after six months she went into remission.
To confirm Gurung’s clean bill of health, Barman recommended that she get a specialized scan at a hospital in southwest New Delhi. A man named Neeraj Chauhan, a longtime administrator across several hospitals’ oncology departments in India, would coordinate the visit, Barman told the family. Piya accompanied her mom to India, where Chauhan assisted them, and sure enough, the scan confirmed Gurung was in remission. But before they returned to Nepal, Piya said, Chauhan and Barman asked them for a favor: Could they carry some medications for other patients across the border?
Piya was suspicious. Her mom was adamant, though, that they follow her doctor’s request, worried that a refusal might affect how he treated her.
After they agreed to the plan, Chauhan delivered the medications to their hotel in a heavily taped Styrofoam box. Despite not knowing what the drugs were or who they were for, Gurung and Piya carried them back to Nepal. They were told there was no need to declare the package at the border. Even then, the arrangement wasn’t that unusual — India and Nepal share an open border, and it’s not illegal to take small quantities of medication across the border for personal use. When Piya and Gurung returned to Nepal, Barman told them via text to refrigerate the package and deliver it to the hospital the next day.
Barman continued to treat Gurung as before, prescribing nivolumab, an immunotherapy medication similar to Keytruda, to prevent the cancer from recurring. But after seven months the cancer returned. Piya said hearing this news was almost worse than learning Gurung’s initial diagnosis, that the remission had felt “too good to be true,” especially after her father’s death. Gurung began chemotherapy again, and then she and Piya returned to India for a second opinion.
Once more, Chauhan and Barman asked them to bring back medication for another patient. This time, Finke, Piya’s husband, sent Barman a stern message expressing his “significant concern” about the request. “To me that was a huge red flag,” said Finke, whose job as an internal medicine doctor at Case Western Reserve University in Ohio made him acutely aware of the power imbalance between doctors and patients in Nepal. “It’s a sense of coercion.”
Chauhan said he would find someone else to do the job or do it himself.
A few months later, Barman prescribed Keytruda, telling the family they had a choice: They could buy each 200 mg dose for 368,000 Nepali rupees, the equivalent of roughly $2,700 at the time, though he didn’t specify from where. Or they could buy it from Chauhan, who “might arrange for a discounted price,” according to WhatsApp messages Piya shared with ICIJ. At first Piya did her own research and found a similar medication that a Nepalese company could import from Bangladesh. She attempted to buy the drugs from that company several times but “customs would keep rejecting it and returning it” without explanation, she said. So instead they turned to Chauhan.

Other patients transported the Keytruda from India to Nepal, Piya said, just as she and her mother had done. The doses arrived sporadically, depending on when people were traveling to and from India. On one occasion, Gurung’s family picked up Keytruda from the Kathmandu airport after Chauhan sent it with a Nepal Airlines passenger.
“We have no idea who this person was,” Piya told ICIJ. “When we met him at the airport, he said he was very worried when they asked him to carry [the medications], but as soon as he heard it could help a cancer patient, he was willing to do it.”
The family ultimately paid Chauhan more than $7,800 for what they were told was Keytruda, Piya said. Another doctor, who collected the payment on behalf of Chauhan, directed them to pay in cash.
“They were very shady about the whole thing,” Piya said, adding that the same doctor asked her to pay him in private, out of sight of anyone else. Chauhan’s lawyer, Ankit Verma, told ICIJ that Chauhan denied that such payments occurred.
Gurung received infusions for two months, but the cancer progressed until she could no longer eat or talk. “She went to sleep, and she was in that state for six or seven days,” Piya said, recalling the last week of her mother’s life. “One of the things I read was they breathe a certain way right before they die. And I saw that breath come, and I went and brought my sister from the other room and said, ‘It’s time.’ So we just stayed around her and just held her hand. And then she passed away.”
The family had Gurung cremated that same night, later spreading some of her ashes in the Bagmati River, a sacred waterway.



Piya moved back to the U.S. and started working again. But her grief was reignited in 2024 when she learned that Chauhan and nearly a dozen others were arrested as part of an alleged cancer drug racket.
New Delhi police said they found 519 empty vials used for cancer drug injections and more than $120,000 in various currencies in Chauhan’s apartment. He later admitted to selling counterfeit drugs, police records show. Chauhan told police he had worked as a manager in the oncology departments of major hospitals in Delhi between 2006 and 2022, according to the records. In 2022, he said, he met the alleged ringleader of the operation, Viphil Jain. Chauhan said he was inspired by Jain, who had described refilling empty vials used in cancer treatments with anti-fungal medicine, which he sold “at half the market price.”
Chauhan and the other defendants are still awaiting trial. Chauhan’s lawyer told ICIJ that the allegations against his client are unfounded and that the police pressured him to make a false confession. He also said that the police fabricated the claims about seizing empty vials and cash from Chauhan’s apartment.
“He didn’t do anything wrong,” Verma said, adding that Chauhan “supplied” authentic medications to Jain, who then altered them without his knowledge. Chauhan also denied asking Gurung’s family to transport medication from India to Nepal, according to Verma.
Barman, the doctor who treated Gurung, told ICIJ in a WhatsApp message that he had “never heard or seen about spurious Keytruda being used anywhere.” He also said that because many drugs were not available in Nepal during the coronavirus pandemic, patients may have carried “medicine for one another during that tough period.” Authorities have not accused Barman of wrongdoing.
During Gurung’s treatment, Keytruda was not — and still isn’t — approved for sale in Nepal. Barman denied medical negligence and said that he “used to send patients to [Chauhan] on their request if something is not available in Nepal.”
“All this,” he said, “was done in good faith and intention to treat.”
A global problem
More than a year after Piya learned about the fake Keytruda from a newspaper article, a newscast in Mexico’s Yucatán Peninsula confirmed Francisco Chávez’s suspicions that his cancer treatment was tainted.
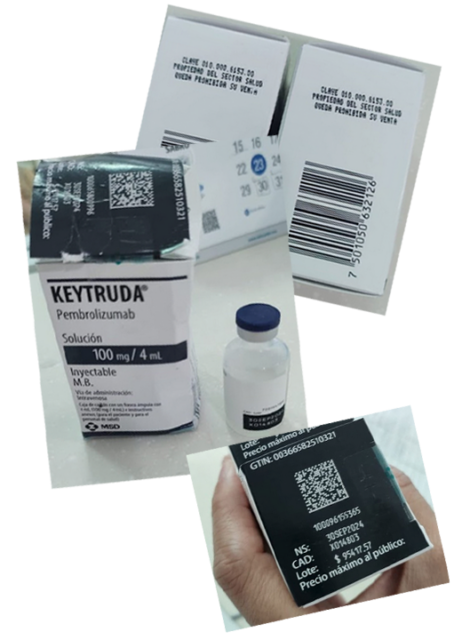
Chávez, an entertainment producer in his 50s, managed tours for musicians — including Ricky Martin and J Balvin — in the town of Mérida. But in late 2022, he lost 123 pounds and his skin began to look gray, he told ICIJ’s partner Univision. He was diagnosed with a cancerous kidney tumor, which was surgically removed, but the cancer had already spread to his lungs. His doctors at the Elvia Carrillo Puerto public hospital prescribed Keytruda, and on his fourth dose he experienced painful side effects: tremors, paralysis and what he described as an out-of-control spike in blood sugar. Chávez took photos of every box of Keytruda he was given.
Within days of his treatment, he filed complaints with three authorities, including the government agency that runs the hospital where he was treated. He demanded to know what was in the vials he was treated with, as well as to see the invoices related to the hospital’s Keytruda purchase. He also asked the hospital to disclose how many patients were affected, but received no response.

Chávez then sent Merck the photos he had taken, along with copies of his complaints. Staff at MSD, as Merck is known outside the U.S. and Canada, traveled to the hospital to collect the suspect material — three product samples with two different batch numbers — and sent them to Merck’s Forensic Services Lab in West Point, Pa., the company told ICIJ. The samples were collected as part of an investigation of the hospital, not in response to Chávez’s photos, Merck said.
Following its analysis, MSD told Chávez in a letter that “certain irregularities were identified that do not correspond to the characteristics of the products manufactured or distributed by our Company.” The letter, signed by the associate director of product integrity for Latin America, said MSD had shared its findings with Mexico’s health regulator, known as Cofepris, and the Attorney General’s Office.
In September 2024, Cofepris issued a warning about counterfeit cancer drugs, including the batch of Keytruda Chávez had reported. It remains unclear how the medication ended up in the hospital.
As Chávez’s treatment continued, he kept recording batch numbers — which turned out to be wise. “I was calmly watching television at home when a nationwide health alert was issued on the news channel, warning about the drug pembrolizumab, known as Keytruda,” he told Univision. He was in disbelief.
The newscast highlighted several fake batch numbers. He went to his phone and checked the photos he had taken. “It turns out that when I see that batch number on television, it’s the one I got,” he said. Chávez realized he had unknowingly been treated with falsified Keytruda a second time but experienced no adverse effects, he said.
Chávez, now 56, said his cancer is “controlled” but his life changed forever after the first unknown substance coursed through his veins. He now suffers from chronic back pain and sudden, inexplicable episodes of paralysis, preventing him from working and undermining his financial security.
“I can no longer move as I used to,” he told Univision. “Before, I would go up and down the stage to put things in place or set up, carry things. All that is over for me now.”
Chávez said he has been gathering information from people who work at the hospital where he was treated as he prepares to file a civil lawsuit. He now believes other patients received suspect medication that, in some cases, caused adverse reactions. When journalists from Univision visited the hospital and asked to speak to the director, staff closed the doors to block access.
While he plans to seek financial compensation, Chávez said he also wants transparency from the hospital and to force the government to investigate. Although he has the lawsuit drafted and ready to file, he said he can’t afford a lawyer to pursue his case. When asked about the high prices of Keytruda set by Merck, Chávez said, “Seeing how you’re wearing your family down financially and physically, that you’re no longer the same person in your daily life and above all, you’re spending a terrible amount of money, and on top of that, [Merck is] profiting off you, it hurts, it hurts a lot.”
In Mexico, counterfeit pharmaceuticals are pervasive because of underfunded public health systems, medication shortages and rampant organized crime. Last year, the U.S. government added Mexico to its priority watchlist of eight countries that have failed to crack down on those “stealing intellectual property.”
Chávez’s case is one of four identified by ICIJ and its media partners in which fake Keytruda was supplied to hospitals in Mexico. At least three of those occurred in state-run institutions. Zook, Merck’s associate vice president for global security, said that MSD has filed 20 criminal complaints related to counterfeit Keytruda with the Mexican Attorney General’s Office.
In December 2021, staff at the Naval Medical Center in Mexico City identified a suspicious-looking vial of Keytruda in a delivery from Top Pharma SA de CV, a company with a yearslong track record of supplying medications to public hospitals and pharmacies. The hospital reported the batch numbers to Cofepris, which sent the vials to MSD and inspected Top Pharma’s headquarters, according to an internal report obtained by ICIJ. Once MSD confirmed the contents were not its patented formula, the regulator issued the country’s first alert on falsified Keytruda in early 2022. Mexico has since issued five more alerts about Keytruda, most recently in March.
In a separate report about the inspection, the regulator said it could not confirm whether Top Pharma had bought the medication from Merck. Instead, the company’s documentation showed it had bought Keytruda from a man who was subsequently added to a public list of substandard distributors as a warning to buyers in the pharmaceutical industry.
ICIJ and its media partners made multiple attempts to contact Top Pharma, which is based in Mexico City. When phone calls, emails and WhatsApp messages went unanswered, a reporter visited the address listed on the company’s website and found only a small pharmacy without Top Pharma signage. A man inside the storefront, who said he was in charge and worked for Top Pharma, refused to answer questions.

A different address, nearly two miles away, is listed as the company’s headquarters in the regulator’s inspection report. The windowless building is about a block from a shopping center in a residential neighborhood. It has no sign or logo, is surrounded by a high fence topped with barbed wire, and is monitored by surveillance cameras.
After the Naval Medical Center incident, Top Pharma was fined more than 240,000 pesos (about $12,800). Yet it has continued to win Mexican government contracts. Public records show Top Pharma has been awarded 91 contracts worth some 42.8 million pesos (about $2.2 million) between 2021 and 2025. In response to questions sent by ICIJ, the navy’s public affairs office said that Top Pharma paid the fine and is legally allowed to keep competing for contracts.
Fighting the fakes
Counterfeit drugs are bad business for pharmaceutical companies. These cheaper alternatives infringe on trademarks, siphon off business, create reputational damage and erode patient trust, all of which can affect a company’s bottom line. To reduce these risks, pharmaceutical companies work with internal and external investigators to hunt them down.
Kris Buckner, the California-based private investigator, has spent 30 years probing everything from knockoff handbags to fake airbag covers. Pharmaceuticals, he said, are the most “egregious” type of product to fake. His company, Investigative Consultants, has worked with Merck on several cases, including a high-profile 2025 investigation that resulted in two Indian brothers being sentenced to prison over a scheme to sell heartburn medication falsely labeled as Keytruda in the United States, according to the U.S. Department of Justice.
Buckner said that he couldn’t comment on the specifics of the investigation but that his company seeks to give law enforcement a “head start.”
Andres Díaz runs a private investigations firm, based in Washington, D.C. — AIT Enforcement — that has helped Merck track down counterfeiters in Latin America. Law enforcement consulted Díaz as part of a 2024 operation in Guadalajara that led to the arrest of “El Tacho,” a man accused of selling counterfeit Keytruda and other drugs. During the raid on El Tacho’s property, Mexico’s navy found 12,500 doses of counterfeit medications, including Keytruda, according to reporting by ICIJ partner El Sol de México. Officials estimated the drugs had a market value of more than 110 million pesos ($5.7 million). The investigation is ongoing, the navy’s public affairs office told ICIJ.

In addition to working with investigators, Merck operates four forensic laboratories to test suspicious products, according to Zook. There, the products are photographed, and their packaging and chemical profiles are compared to authentic Keytruda. Merck’s forensic labs tested more than 800 product samples in 2024, according to the company’s website. That included nine Keytruda samples from the New Delhi operation allegedly involving Chauhan. Eight did not contain the drug’s active ingredient, pembrolizumab, according to Merck’s analysis.
When falsified Keytruda is identified, “Merck works closely with the relevant law enforcement agencies and health authorities worldwide, sharing intelligence, forensic evidence and operational support to enable them to conduct criminal and administrative enforcement actions that will protect public health,” Zook said. “Where applicable, Merck will also pursue civil legal action against those involved in the manufacturing and distribution of falsified medicines.”
Merck also actively monitors suspicious online listings on social media sites and e-commerce platforms like Amazon, eBay, Mercado Libre and IndiaMART. The company reported that it helped remove around 30,000 listings in 2024.
Stamping out dangerous imitations has become increasingly challenging, however, not just for pharmaceutical companies, but also for law enforcement. Today’s criminal networks use encrypted messaging apps to reach customers, hide their identities and wealth through complex financial networks, and abuse the international mail system to misdeclare shipments, a spokesperson for the U.S. Food and Drug Administration said in a statement.
In multiple interviews, experts told ICIJ that pharmaceutical companies and policymakers should do more to address the underlying problems that cause these fakes to flourish in the first place: pricing and availability.
“There’s a lot of evergreening and other patent issues that go on at the end of the life of a product,” said Mackey, the UC San Diego professor. Evergreening is a strategy pharmaceutical companies use to create a monopoly around their drug by filing patents for minor modifications such as dosing amounts and frequency. Keytruda’s key patents were set to expire in 2028, but ICIJ found that Merck has continued to obtain additional patents for the drug beyond its active ingredient, potentially making it harder for competitors to enter the U.S. market for another 14 years.
Saifuddin Ahmed, principal investigator for BESAFE, a Johns Hopkins University initiative targeting the spread of dubious medicines, said that publicly funding the research and development of drugs like Keytruda could bring down prices. “If the drug is developed by the public-funded projects, that drug possibly will be distributed at a mass transport, cheaper rate,” he said.
For now, inaccessibility continues to plague health care systems worldwide, leaving many patients to fend for themselves. “Patients need access to affordable, therapeutic drugs that are authentic, that are going to save their lives,” Buckner said. “That’s the bottom line.”
‘The worst thing’
To remember her mother each year, Bhinnata Piya donates to a food-related charity. But that tradition is far from bringing her closure.
“How do you even process something like this?” she said of possibly giving her mother fake drugs. “I think once maybe we know that this won’t happen to others and they’re held accountable, maybe I can close that process mentally and move on.”

In an effort to do that, she filed a medical negligence lawsuit in Nepal against Barman, Chauhan, the doctor who accepted her Keytruda payments and the hospital her mother was treated in. Because Chauhan is not a Nepalese citizen, the case is moving slowly.
Francisco Chávez, too, continues to feel haunted by his experience as he works to get his case off the ground.
“I think profiting from people’s health,” Chávez said, “is the worst thing that can happen in the world.”
Contributing reporters: Angie Sandoval and Gerardo Reyes (Univision), Violeta Santiago (Quinto Elemento Lab), Aldo Canedo (El Sol de México), Carlos Carabaña (El País), Denise Ajiri, Jelena Cosic and Karrie Kehoe (ICIJ)