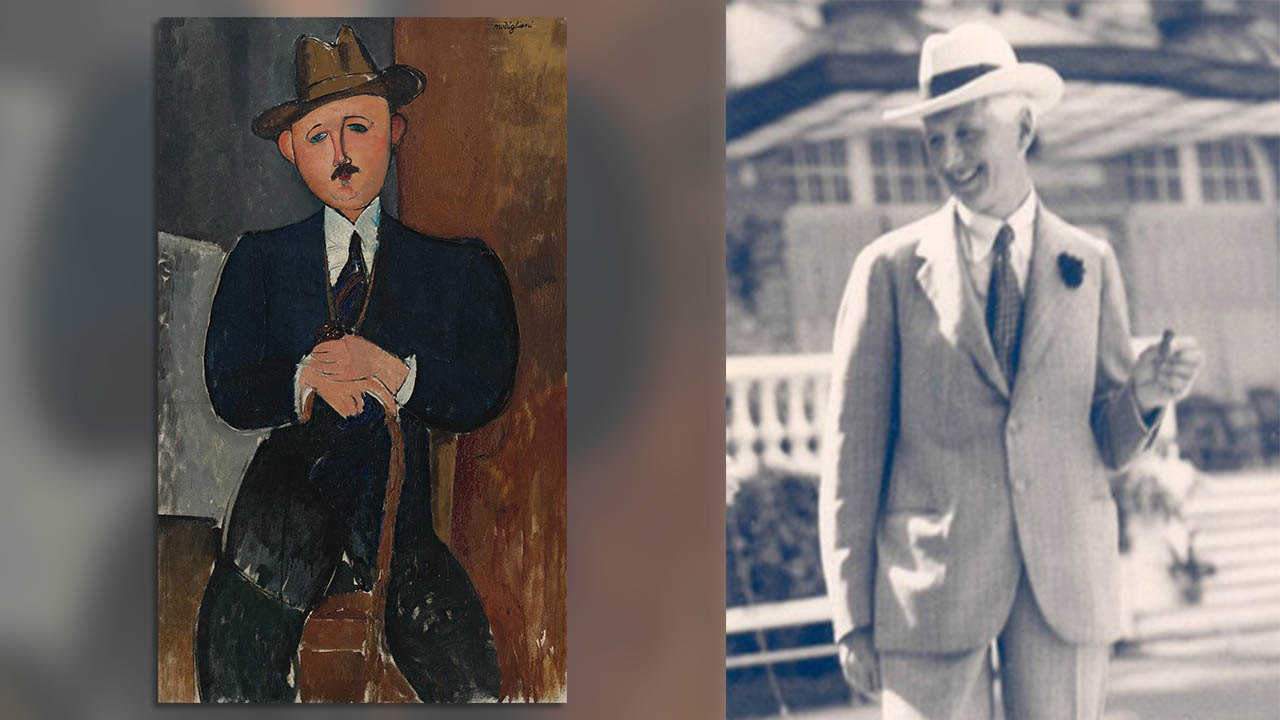Impact
ICIJ has redefined what investigative journalism looks like in the 21st century. We have repeatedly conducted journalistic collaborations so big, so visionary and so aspirational that they are unlike any others in history.
We have led the biggest cross-border journalism projects in history, including the Panama Papers and Pandora Papers. Our investigations have shaken the establishment and led to public protests, multiple arrests, sweeping legal reforms, and official inquiries in dozens of countries.
May 19, 2026
Crypto ATM operator Bitcoin Depot files for bankruptcy
With regulators cracking down on the cash-to-cryptocurrency kiosks in a bid to stop scammers, Bitcoin Depot has said that its business model is no longer viable.
May 17, 2026
Alleged cryptocurrency Ponzi scheme ‘goddess’ extradited from Thailand to face conspiracy charges in US
Authorities say Olena Oblamska was one of the founders of Forsage, a cryptocurrency platform allegedly used as part of a global pyramid scheme.
Apr 28, 2026
‘Escalating efforts’: A year after China Targets, Beijing’s global campaign against dissenters continues
In 2025, China was the most prolific perpetrator of transnational repression, a new Freedom House report found.
Apr 24, 2026
Former co-owner of Panama Papers law firm convicted of aiding and abetting tax evasion
Over a decade after the ICIJ investigation, a German court found the Swiss lawyer guilty of enabling a tax loss of about $15 million.
Apr 23, 2026
‘Unacceptable’: Lawmakers react to revelations from ICIJ’s Cancer Calculus investigation
In hearings around the world, politicians and experts called for inquiries into inequities surrounding Merck's anti-cancer drug Keytruda.
Apr 07, 2026
Global headlines and a public reckoning: Ten years of the Panama Papers, part 3
On the 10-year anniversary of the Panama Papers, journalists and a Nobel-wining economist share their recollections of how the story unfolded, and how the investigation still resonates today.
Apr 06, 2026
Judge orders Nazi-looted Modigliani linked to Panama Papers be returned to heirs
The estate of a Jewish art dealer has won a decade-long court battle over a $25 million painting whose ownership was exposed by secret financial documents obtained by ICIJ and its media partners.
Apr 02, 2026
WATCH: The Panama Papers — ten years of impact
A decade after the Panama Papers made headlines around the world, the investigation remains a milestone moment in the global push for financial equality and transparency.
Apr 02, 2026
Behind the veil of secrecy: Ten years of the Panama Papers, part 2
On the 10-year anniversary of the Panama Papers, journalists recall how it all happened, and how the investigation took the world by storm.
Apr 02, 2026
Ten years after the Panama Papers, enablers and tax cheats are still being brought to justice
A look back at a decade of changes after the Pulitzer-Prize winning investigation sent a shock through the offshore world.
Mar 31, 2026
The story that rocked the world: Ten years of the Panama Papers, part 1
Ten years after the Panama Papers hit front pages around the world, ICIJ unpacks how the groundbreaking investigation came together, beginning with an unprecedented data leak.
Mar 24, 2026
Canada revokes dozens of crypto firms’ registrations
Reporting by The Toronto Star as part of ICIJ's Coin Laundry investigation found clusters of crypto services operating unlawfully.
Mar 12, 2026
Human rights court calls on governments to crack down on weapons trafficking
The Inter-American Court of Human Rights said that governments have a duty to more closely monitor firearms manufacturers and put a stop to illegal trafficking.
Feb 26, 2026
Greek court convicts Intellexa founder Tal Dilian, three others in wiretapping scandal
The former Israeli intelligence officer’s spyware has helped some of the world’s most brutal regimes spy on journalists and political opponents.
Feb 26, 2026
Massachusetts sues Bitcoin Depot, alleging the crypto ATM operator knowingly facilitated crypto scams
The Massachusetts Attorney General’s lawsuit is the latest in a series of state legal actions against one of the world’s largest crypto ATM operators.
Feb 10, 2026
Nearly half of powerful .50-caliber ammo seized by Mexican government came from US Army plant, defense minister says
Mexican officials shared the data in response to an investigation by ICIJ and media partners.
Feb 06, 2026
Former Nigerian oil minister stands trial in the UK on bribery charges
Prosecutors accused former minister Diezani Alison-Madueke, who featured in the ICIJ-led Panama Papers investigation, of living a “life of luxury in London” fueled by corrupt Nigerian oil deals.
Feb 05, 2026
Canada names first foreign interference watchdog
Canadian parliament voted to create the position over a year and a half ago to counter a rising threat of transnational repression.
Feb 04, 2026
Beijing’s backtrack on Xinjiang detention camps spurred by ICIJ investigation, research finds
Academic research shows that investigative reporting, NGO advocacy, and scholarly scrutiny pushed Chinese authorities from denial to dismantling parts of their mass detention system for Uyghurs.
Jan 29, 2026
New EU report urges more aggressive action against transnational repression
The report cites ICIJ’s China Targets investigation and calls for improved coordination across the bloc.
Dec 09, 2025
Cambodian payment processor freezes customer funds before regulators shut it down
Huione Pay, a key arm of Huione Group spotlighted in ICIJ’s Coin Laundry investigation, has been closed by Cambodia’s national bank and its assets liquidated.
Oct 28, 2025
Dartmouth researchers use ICIJ database to show how the ultrawealthy exploit the offshore world
ICIJ spoke to author Herbert Chang about the study, which found that elites in both democratic and authoritarian countries are well-versed in financial secrecy tactics.
Oct 15, 2025
New UN report highlights China’s alleged targeting of human rights activists
The report, which recounts recent reprisals from two dozen countries, underscores ICIJ’s reporting on how Beijing abuses international institutions in its campaign to silence critics abroad.
Sep 17, 2025
FinCEN plans to delete data on U.S. companies from beneficial ownership database
Five years after ICIJ published the FinCEN Files, the Trump administration has walked back significant anti-money laundering rules.
Sep 12, 2025
Chelsea FC faces over six dozen charges for improper conduct under Abramovich’s ownership
The Football Association announced it is investigating payments routed through offshore companies that were uncovered as part of ICIJ’s Cyprus Confidential investigation.